CONTROL study results show improved tolerability of NERLYNX® (neratinib) with all the investigated diarrhoea prophylaxis strategies | Pierre Fabre Pharma Norden AB

Puma Biotechnology and Pierre Fabre Enter into Exclusive License Agreement to Develop and Commercialize NERLYNX® (neratinib) in Europe | Business Wire

ESMO 2017: Effects of neratinib (N) on health-related quality of life (HRQoL) in early-stage HER2+ breast cancer (BC): longitudinal analyses from the phase III ExteNET trial
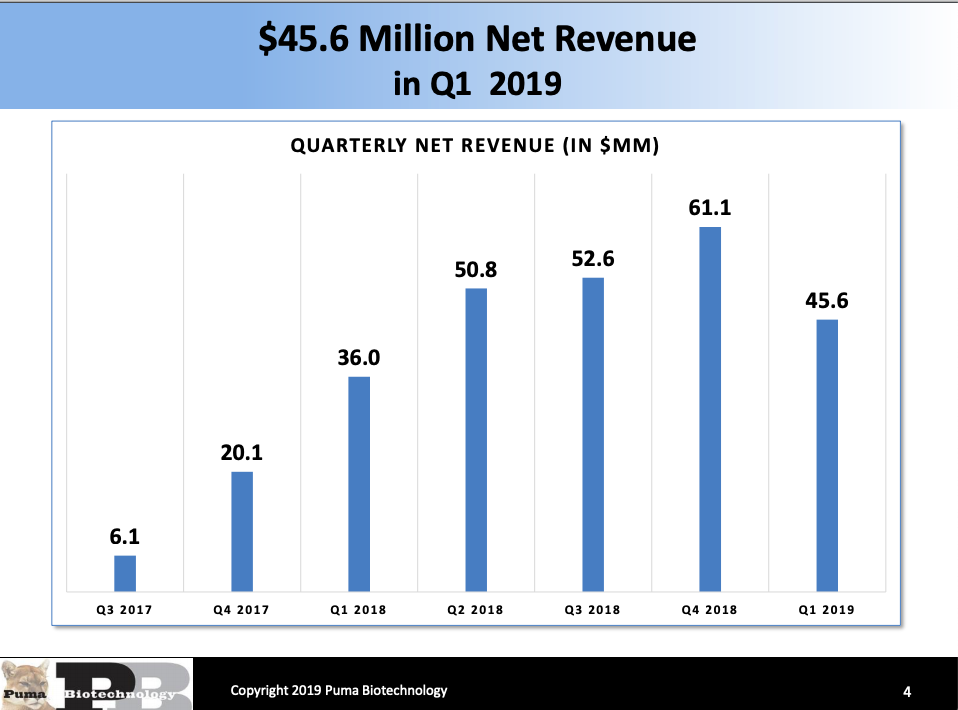
Puma Biotechnology: Nerlynx Peak Sales Estimates Have Come Down, But The Intense Selling Still Makes This A Buy (NASDAQ:PBYI) | Seeking Alpha

PharmaShots Interview: Pierre Fabre' Roberta Valenti Shares Insights on Nerlynx (neratinib) for the Treatment of HER2 Early Breast Cancer